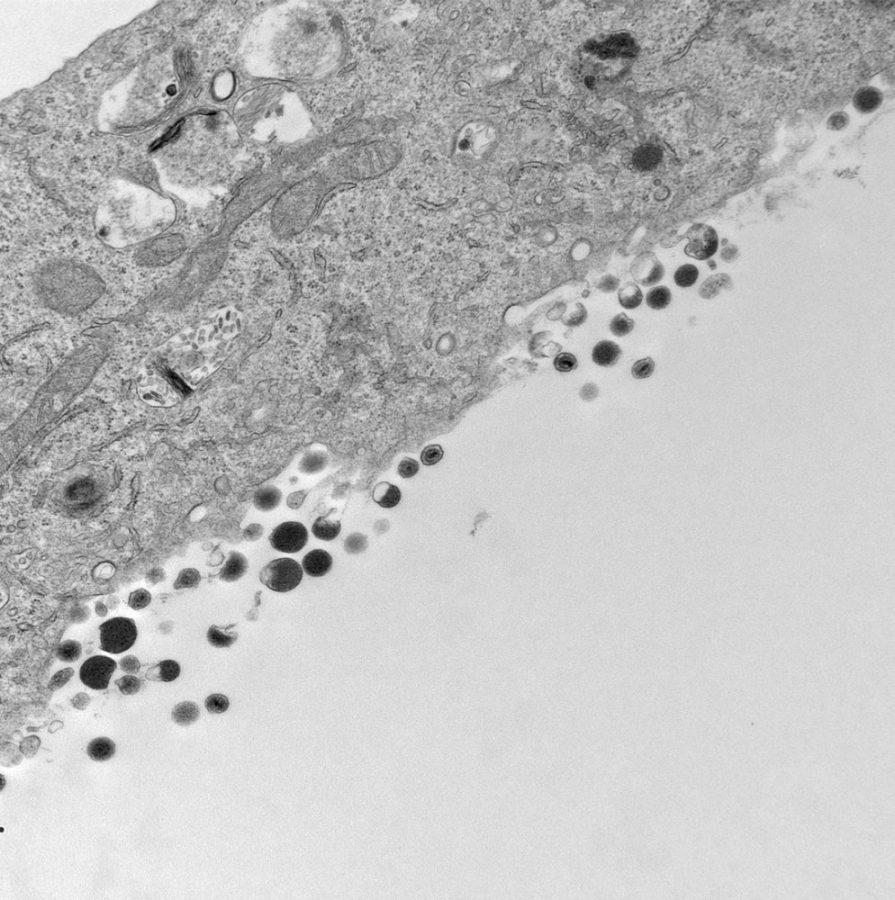
Cytomegalovirus, or CMV, is one of eight human herpes viruses that are known for causing latent infections — infections that don’t exhibit symptoms — and 60 to over 90 percent of the population is already infected with CMV. The Goodrum Lab at the UA is seeking to determine how this virus works genetically and its effects on congential infections, where an infection in the mother can affect a newborn or fetus.
CMV is unique in that it can cause a congenital infection that affects one in 150 births in America. More severe cases of congenital CMV are thought to occur when a seronegative woman, or a woman who has not been infected and does not have antibodies for the virus, is exposed to the virus for the very first time.
“The baby of a seronegative woman is at risk for severe congenital infection if she were to contract the virus during her pregnancy, because the virus has time to move across the placenta before there’s any immune response to hold it back,” said Felicia Goodrum, associate professor for the department of immunobiology. “To acquire an immune response to CMV that would be protective takes weeks to develop after infection.”
Goodrum’s lab is currently studying a gene locus UL133-UL138 and has found that these viral proteins are responsible for both suppressing and promoting viral replication, which is why the virus is able to sustain a persistent, latent infection.
Katie Caviness, a genetics graduate student, focuses on the gene UL136, which codes for five different proteins. The functions of these proteins are not yet known, but using genetics, Caviness has determined that two of the proteins suppress viral replication and two of the proteins promote replication.
To determine how the UL136 proteins function, Sebastian Zeltzer, a graduate student studying cellular and molecular medicine, is studying how the proteins encoded by the gene UL136 interact with host cell proteins, which are proteins expressed by the human cells. These host proteins are important for embryonic development, cancer, tissue regeneration and organogenesis. Therefore, the interactions between UL136 and host proteins may be important to disease associated with the congenital infection.
“In development, when the pathways targeted by UL136 get turned on, cells proliferate and migrate,” Zeltzer said. “This is important for the establishment of organs and what are called the axes of development. If the pathways are turned off, it will kill the developing embryo, but if it gets turned on to an even greater extent, that can lead to some pretty terrible mutations as well. We have some evidence that the virus is actually turning it up instead of turning it down.”
A reservoir of latent virus within the body are bone marrow hematopoietic progenitor cells, which are precursors to the body’s blood and immune cells.
“It achieves a latent infection in these cells and does not make new virus, so it just totally hangs out,” said Mike Rak, a graduate student studying cellular and molecular medicine. “Very few viral proteins are expressed. The immune system can’t target these latently infected cells. The immune system can only target the cells that express viral proteins.”
This also makes it very hard for antivirals as well, which can only target cells actively replicating the virus.
In immunocompromised patients, such as cancer patients, organ transplant recipients and HIV/AIDS patients, CMV can infect every cell in the body, which can lead to a variety of symptoms such as blindness, gastroenteritis and pneumonia.
“When you lose your immune system to HIV infection or to suppression following organ transplantation, the virus can reactivate, and that may ultimately cause disease,” Goodrum said.
The Goodrum Lab collectively works to understand how CMV contributes to and controls its persistence within the human host.
“Our hope is that our work will ultimately identify strategies to prevent disease in the immunocompromised or prevent birth defects in the congenital infection,” Goodrum said. “Understanding the basic biology of CMV infection and latency are paramount to achieving this goal.”
_______________
Follow Connie Tran on Twitter.