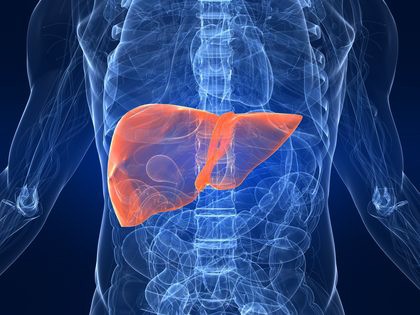
A team of researchers at the UA College of Pharmacy recently discovered a new molecular pathway that sheds light on an important protein that could help slow or reverse the progression of end-stage liver disease.
The researchers discovered a new regulatory pathway that involves the protein Nrf2, which plays a significant role in fighting “oxidative stress” in the body. Nrf2 is found in almost every cell. It helps the body clean out free radicals, which are very reactive molecules that cause damage to the body and trigger oxidative stress in the cells, said Tongde Wu, a UA graduate who worked on the project for her dissertation.
Wu was attempting to identify the stress pathways involved in end-stage liver cirrhosis by comparing samples of normal and cirrhotic liver tissue when she noticed that there were decreased levels of Nrf2 in the cirrhotic livers.
What’s more surprising is that the protein that normally inhibits the amount of Nrf2 in the body, called Keap1, was deactivated, said Donna Zhang, a professor in the department of pharmacology and toxicology who was a primary researcher in the study.
According to Wu, the low Nrf2 levels should not have occurred; there is significant oxidative stress in cirrhotic livers, so they should have been producing higher levels of the protein. She began to wonder what was going on to inhibit the Nrf2 production, and wanted to see if there was a way to recover the function of the protein to possibly rescue cirrhotic livers.
When a person has a cirrhotic liver, the cells in the liver are experiencing multiple types of stress. One type is oxidative stress, caused by the glut of free radicals. Another type of stress is called Endoplasmic Reticulum stress, which is caused by the overproduction of damaged proteins in a cell. During ER stress, the body inhibits the production of Nrf2, as the energy is being spent to combat ER stress rather than the oxidative stress caused by free radicals, Wu said.
It turns out that the ER stress in a cirrhotic liver triggers a second protein involved in the regulation and degradation of the Nrf2, called Hrd1. According to Eli Chapman, an assistant professor in the department of pharmacology and toxicology who contributed to the research, Hrd1 levels increase when there is cellular stress to help deal with the damage, but higher levels of Hrd1 mean further degradation of Nrf2.
While researchers knew that Keap1 could degrade Nrf2 in this manner, they had no idea that the Hrd1 protein could also destroy Nrf2. This shows a molecular connection between the ER stress and the oxidative stress pathways, Zhang said.
“Keap1 has a similar characteristic as Hrd1, as they both send Nrf2 to degradation,” Wu said. “When ER stress is predominant in the cells, it activates the unfolded protein response, which activates Hrd1, which sends Nrf2 for degradation. The ability of the cells to handle oxidative stress is suppressed, which means even more damage to the cells.”
The discovery of this second protein triggered in the ER stress pathway could prove highly useful for therapies to combat and reverse the progression of liver disease.
“We show if you are able to inhibit Hrd1 and prohibit the degradation of Nrf2, it alleviates liver cirrhosis,” Zhang said.
Follow Michaela Kane @MichaelaLKane